Listeria food alert: evolution of cases
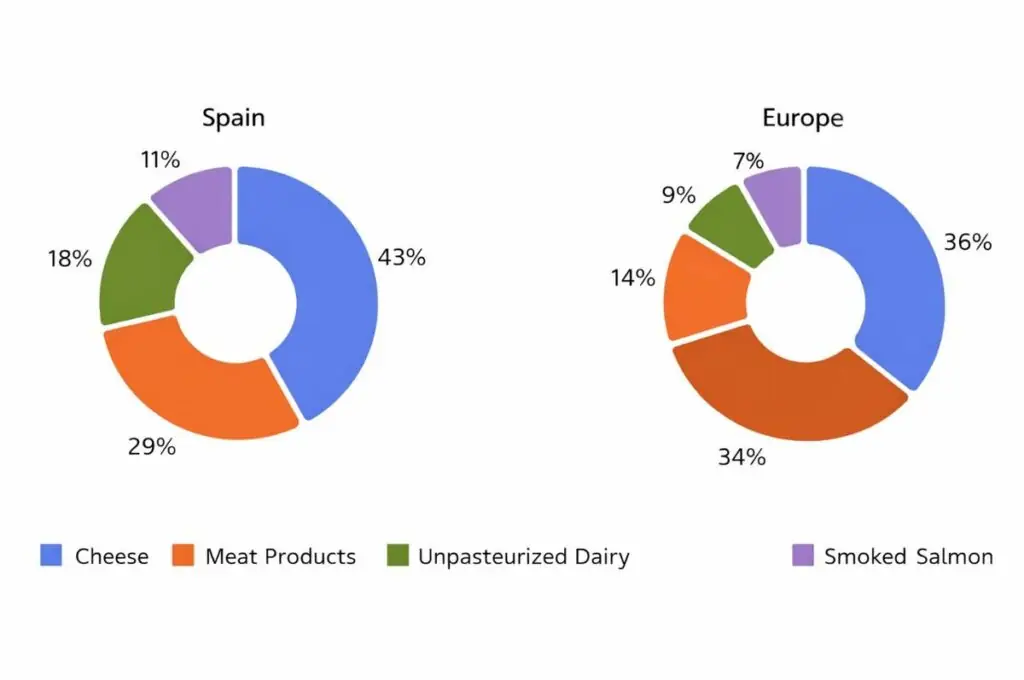
Between 2020 and 2025, multiple incidents related to the presence of Listeria monocytogenes in food have occurred, as well as outbreaks of listeriosis that have affected public health in different countries.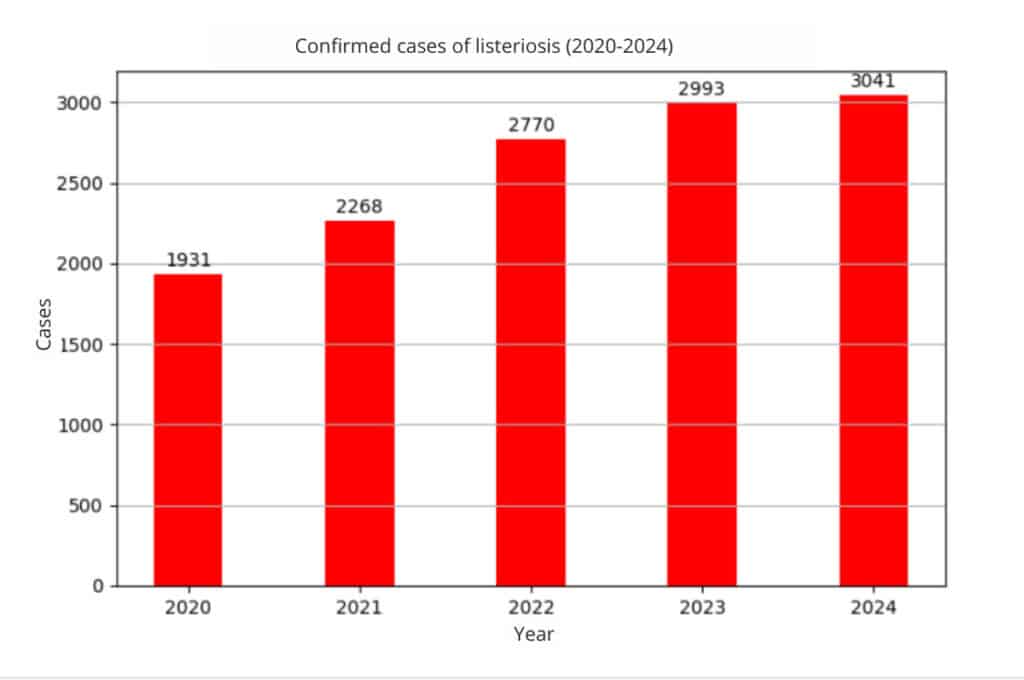 This demonstrates that Listeria monocytogenes remains a priority pathogen in microbiological surveillance, especially in:
This demonstrates that Listeria monocytogenes remains a priority pathogen in microbiological surveillance, especially in:
- Soft cheeses
- Smoked fish
- Ready-to-eat meat products
What does European legislation require?
The amendment of Regulation (EU) 2073/2005 through Regulation (EU) 2024/2895 responds to the need to guarantee consumer protection throughout the entire shelf life of the product, to the evidence of Listeria monocytogenes growth outside the direct control of the producer and to the need to establish uniform microbiological criteria across the entire food chain. It introduces important changes in food safety criteria, particularly for ready-to-eat foods:| Microbiological criterion | Application |
|---|---|
| Absence in 25 g | Mandatory when it cannot be demonstrated that Listeria will not exceed 100 cfu/g during shelf life |
| ≤ 100 cfu/g | Only applicable if the operator scientifically demonstrates that growth is controlled |
How to address the risk of Listeria monocytogenes?
New consumption trends, together with regulatory changes, require the food industry to adopt a rigorous and multifactorial approach to prevent the development of Listeria monocytogenes, especially in ready-to-eat foods. To achieve this, action must be taken on several fronts:- Establish, validate and verify the shelf life of the food.
- Review formulations.
- Environmental and biofilm control.
- Advanced analytical methods.
- Documentation management and HACCP.
- Staff training.
Tools available to validate shelf life
Shelf-life validation in ready-to-eat foods requires specific tools that allow the evaluation of microbiological risk. Among the most relevant are growth potential studies. To carry them out, the technical guidelines of the European Union Reference Laboratory for Listeria monocytogenes (EURL Lm) are followed, as set out in its technical document on challenge tests and durability studies. This protocol provides a scientific basis for designing studies that simulate real storage conditions, evaluate the behaviour of Listeria monocytogenes in different food matrices and determine whether the food can be considered safe throughout its entire shelf life. The objective of Listeria monocytogenes growth potential studies is to evaluate whether this microorganism can develop in a ready-to-eat food during its shelf life.