Studies and other services
- R&D&I Programmes
- Technological Services
- Consultancy Services
- Laboratory Services
- Training and Events
- Sectors
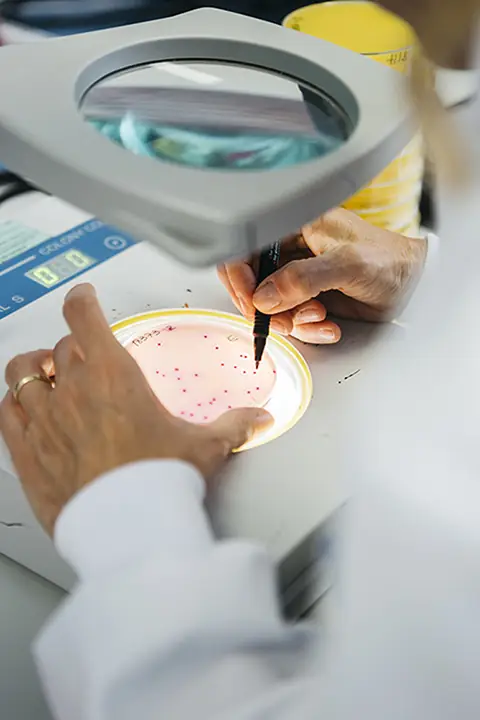
Our solutions include:
| Responsible | AINIA |
| Address | Calle Benjamín Franklin, 5 a 11, CP 46980 Paterna (Valencia) |
| Purpose | To attend to, register and contact you to resolve the request you make to us through this contact form |
| Legitimation | Your data will be processed only with your consent, by checking the box shown on this form |
| Recipients | Your data will not be transferred to third parties. |
| Rights | You have the right to request access to, correct or delete your data. You can also request that we limit its processing, oppose it and request the portability of your data by contacting our postal address or [email protected] |
| More info | You can find more information in our Privacy Policy |
| DPO | If you have any questions about how we will treat your data or would like to make any suggestions or complaints, please contact the Data Protection Officer at [email protected] or at the Data subject support form |
I consent to the use of my personal data to process my request, as set forth in your Privacy Policy.
I consent to the use of my data to receive information and commercial communications from your entity.
